Bacteriostatic Water 30ml – Uses, Dosage & Buying Guide | Bacteriostatic Water USA
19 Mar, 2026
2750 Views 0 Like(s)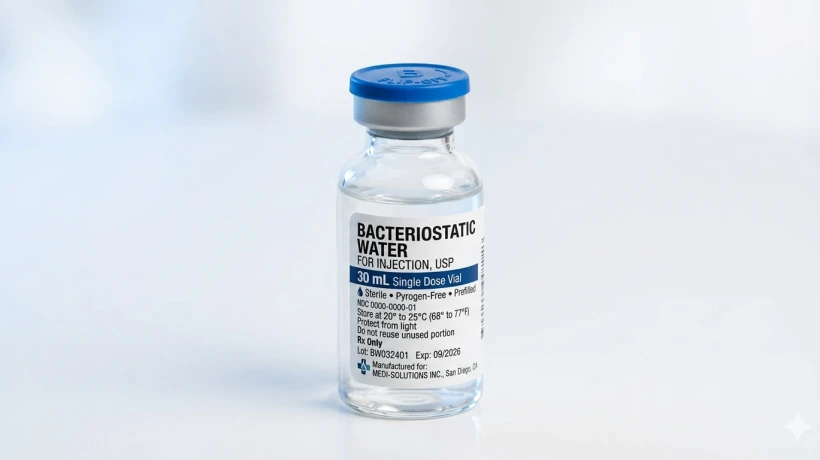
Looking for bacteriostatic water 30ml? Learn what it is, how it's used for reconstitution, storage tips, and what to look for when buying USP-grade quality.
If you've spent any time researching peptide reconstitution, hormone therapies, or compounding practices, you've likely come across the term bacteriostatic water. And if you're here, chances are you're specifically looking at the 30ml size — one of the most practical and widely used volumes available on the market today. Whether you're new to the concept or just looking to deepen your understanding, this guide will walk you through everything that matters: what bacteriostatic water is, why the 30ml size is preferred by so many, how it's used correctly, and what to look for when purchasing it.
Let's break it all down.
What Is Bacteriostatic Water?
Bacteriostatic water is sterile water that contains 0.9% benzyl alcohol as a preservative. That preservative is what makes it fundamentally different from regular sterile water or saline. The benzyl alcohol doesn't kill bacteria outright — instead, it inhibits bacterial growth, which is precisely where the name comes from. "Bacteriostatic" literally means "stopping bacteria."
This distinction matters enormously in practice. Because bacteriostatic water resists contamination over repeated needle entries, it can be used multiple times from a single vial — unlike standard sterile water, which should generally be discarded after a single use. This multi-dose capability makes it the go-to diluent in a wide range of clinical and research settings.
Bacteriostatic water is used to reconstitute (dissolve) lyophilized (freeze-dried) substances such as peptides, hormones like Human Growth Hormone (HGH), certain vaccines, and other injectable compounds. Without a proper diluent, these substances simply cannot be administered safely or effectively.
Why the 30ml Size Is the Sweet Spot
You'll find bacteriostatic water available in several vial sizes — typically 10ml, 20ml, and 30ml. So why does the 30ml option come up so often in conversations about reconstitution?
The answer is practical efficiency. A 30ml vial gives you more diluent per purchase, which means fewer vial changes, less handling risk, and better cost-effectiveness — especially if you're managing ongoing reconstitution needs. For those working with multiple peptides or higher-volume research protocols, the 30ml size reduces the frequency of opening new vials, which in turn reduces the chances of contamination.
At the same time, 30ml strikes a balance. It's not so large that the preservative becomes overwhelmed or the vial gets too unwieldy. Most researchers and medical professionals find that a 30ml vial lasts a reasonable amount of time without compromising sterility — provided proper technique is used every time.
In short: the 30ml size is enough to be convenient, small enough to remain safe, and common enough that it's widely stocked.
How Bacteriostatic Water Is Used for Reconstitution
Reconstitution is the process of mixing a lyophilized powder with a liquid diluent to produce an injectable solution. Here's a general overview of how bacteriostatic water fits into that process:
Step 1 — Gather your materials. You'll need your lyophilized vial, a 30ml vial of bacteriostatic water, an appropriate syringe, alcohol swabs, and sterile technique throughout.
Step 2 — Clean everything. Wipe the rubber stoppers on both vials with an alcohol swab and let them air dry. Never skip this step — it's your first line of defense against contamination.
Step 3 — Draw the bacteriostatic water. Insert the syringe needle into the 30ml vial and draw the required volume of bacteriostatic water. The amount you need depends on the specific compound you're reconstituting and the concentration you want to achieve.
Step 4 — Add slowly to the lyophilized vial. Inject the bacteriostatic water gently down the side of the vial — never directly onto the powder. This gentle approach preserves the compound's integrity.
Step 5 — Mix carefully. Swirl the vial slowly. Do not shake vigorously, as this can denature sensitive proteins and peptides. Allow the powder to dissolve fully before use.
Step 6 — Store correctly. Once reconstituted, most solutions should be refrigerated at 2–8°C and used within a specified timeframe — typically 28 to 30 days, depending on the compound.
The multi-dose nature of the 30ml vial of bacteriostatic water means you can return to it for each reconstitution without replacing it every time — as long as proper aseptic technique is maintained.
Key Things to Look for When Buying Bacteriostatic Water 30ml
Not all bacteriostatic water is created equal. When you're making purchasing decisions, here are the factors that genuinely matter:
USP-Grade quality. Always look for bacteriostatic water that meets United States Pharmacopeia (USP) standards. This means the water has been tested for sterility, pyrogens, and proper benzyl alcohol concentration.
Packaging integrity. The vial should be sealed with a tamper-evident cap. Any sign of damage, leakage, or compromised seals is a dealbreaker.
Clear labeling. The label should clearly list the ingredients — specifically that it contains 0.9% benzyl alcohol — along with lot number, expiration date, and manufacturer information.
Reputable sourcing. This is non-negotiable. You want to buy from a trusted supplier who can verify the quality and purity of their product. Bacteriostatic Water USA is one example of a supplier that focuses specifically on providing USP-grade bacteriostatic water with transparent sourcing and quality standards — important when you need something you can actually trust.
Expiration date. Never use bacteriostatic water past its expiration. The preservative efficacy degrades over time, and using expired product puts both the compound and the recipient at risk.
Storage and Handling Tips
Bacteriostatic water should be stored at controlled room temperature — generally between 20°C and 25°C — before opening. Once opened and in use, refrigeration is often recommended, though always defer to the manufacturer's guidelines.
A few habits that make a real difference in maintaining sterility:
Always use a new, sterile needle each time you draw from the vial. Never reuse needles, even if you're drawing from the same vial. Avoid touching the rubber stopper with ungloved hands. Keep the vial out of direct sunlight and away from extreme temperatures. If the solution ever appears cloudy, discolored, or contains visible particles — discard it immediately.
These habits aren't overcautious. They're what separates a safe, effective protocol from a compromised one.
Common Misconceptions About Bacteriostatic Water
"It's the same as saline." Not quite. Saline (0.9% sodium chloride) is also used as a diluent, but it doesn't contain a preservative. This makes saline a single-use product in most contexts, while bacteriostatic water supports multiple draws from the same vial.
"More benzyl alcohol means better preservation." The 0.9% concentration is specifically calibrated for safety and efficacy. Higher concentrations of benzyl alcohol can be toxic — particularly in neonates. Always use pharmaceutical-grade bacteriostatic water with the standard concentration.
"Any sterile water will do." Regular sterile water lacks the preservative that inhibits contamination over time. Using it in a multi-dose setting dramatically increases the risk of bacterial growth in the solution.
Frequently Asked Questions (FAQs)
Q: How long does bacteriostatic water last after the vial is opened? A: Generally, an opened vial of bacteriostatic water remains usable for up to 28 days when stored properly and handled with sterile technique. Always check the manufacturer's specific guidelines and never use it past the printed expiration date.
Q: Can bacteriostatic water be used for all injectable compounds? A: Not always. While it's suitable for reconstituting most peptides and hormones, some compounds may require sterile water for injection (SWFI) or another specific diluent. Always follow the reconstitution instructions provided with your specific compound.
Q: Is bacteriostatic water safe for intramuscular and subcutaneous injections? A: Yes, bacteriostatic water is widely used for both intramuscular (IM) and subcutaneous (SQ) injections when reconstituting appropriate compounds. However, it should not be used in large volumes intravenously, as the benzyl alcohol concentration can be problematic.
Q: Why is 30ml preferred over 10ml vials? A: The 30ml size offers greater volume per vial, reducing how often you need to open a new vial. This means less handling, lower contamination risk, and better overall convenience — especially in research or clinical settings with ongoing needs.
Q: Can bacteriostatic water be used for neonates or infants? A: No. Benzyl alcohol has been associated with serious adverse effects in neonates. Bacteriostatic water should never be used in newborns or very young infants. Sterile water for injection without preservatives is the appropriate choice in those cases.
Q: How do I know if my bacteriostatic water has been compromised? A: Look for cloudiness, discoloration, visible particles, or any unusual smell. A compromised seal or damaged vial is also cause for immediate disposal. When in doubt, discard and use a new vial.
Q: Does the 30ml vial need to be refrigerated? A: Before opening, store at controlled room temperature as directed by the manufacturer. After opening, refrigeration may extend the product's usable life, but always defer to the specific storage instructions on your vial.
Final Thoughts
Bacteriostatic water 30ml isn't a glamorous product — it's a supporting player. But like all supporting players in a well-run operation, it matters more than people often realize. The difference between a correctly reconstituted compound and a contaminated one often comes down to diluent quality, proper technique, and appropriate vial size.
Understanding what you're working with, why the 30ml size earns its reputation, and how to store and handle it correctly puts you in a far stronger position — whether you're a researcher, a healthcare professional, or someone navigating a prescribed protocol under medical supervision.
Choose quality. Follow sterile technique. Store properly. These aren't just suggestions — they're the foundation of safe and effective reconstitution practice.


Comments
Login to Comment